Arq Bras Cardiol: Imagem cardiovasc. 2025; 38(1): e20240124
First Percutaneous Orthotopic Tricuspid Valve Implantations using the LuX-Valve Plus Device in Latin America: Report of the First Three Cases
DOI: 10.36660/abcimg.20240124i
Abstract
Background:
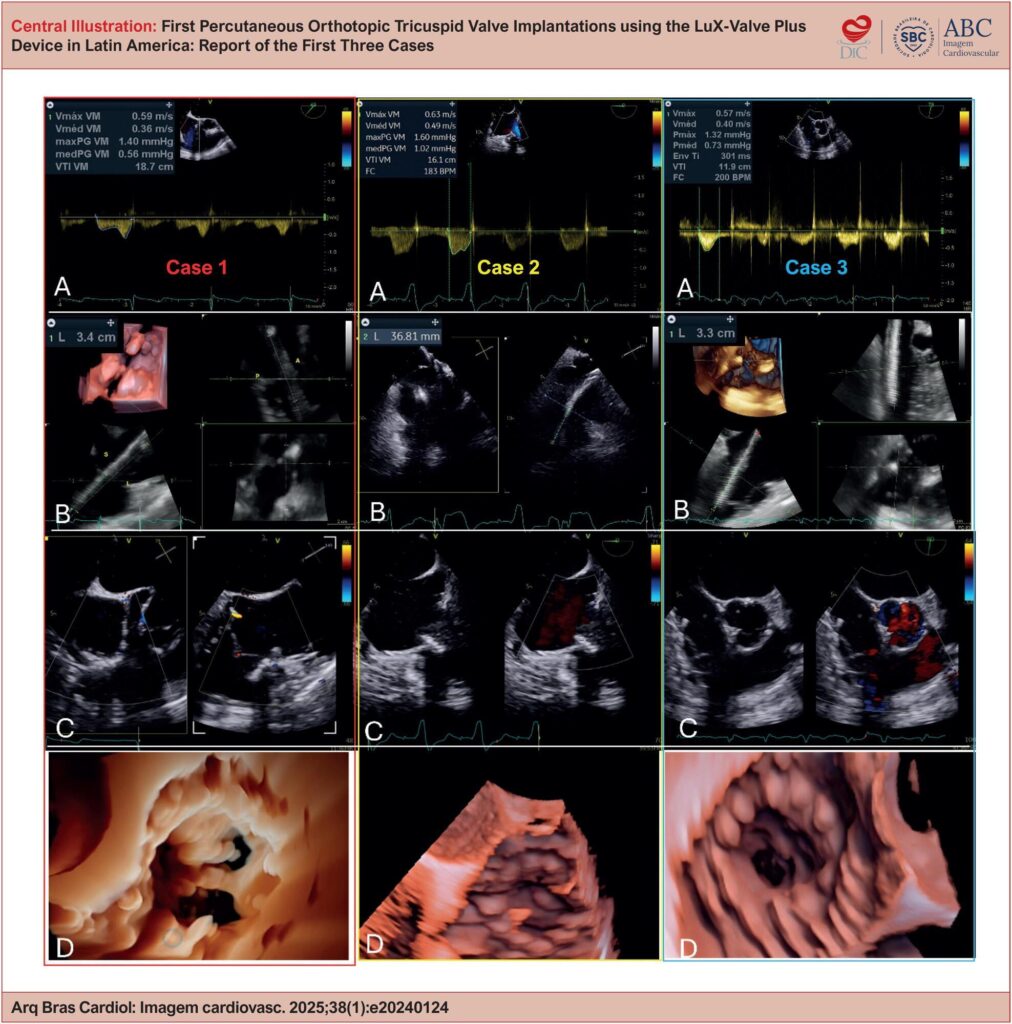
Severe tricuspid regurgitation (TR) is associated with high morbidity and mortality, especially in patients with contraindications for surgery. Transcatheter tricuspid valve replacement has emerged as a minimally invasive alternative for this population, with the LuX-Valve Plus device designed for orthotopic implantation via the transjugular route.
Objective:
To report the first 3 cases of transcatheter tricuspid valve replacement using the LuX-Valve Plus device in Latin America, assessing the feasibility, safety, and immediate clinical outcomes.
Methods:
The study included 3 female patients with torrential TR and high surgical risk. Pre-procedure assessment included transesophageal echocardiography, 3-dimensional transthoracic echocardiography (3D TEE), computed tomography, and cardiac catheterization. The procedure was performed via transjugular access, under general anesthesia, with 3D TEE and fluoroscopy guidance. Implant efficacy was assessed as resolution of TR, transprosthetic gradient, and post-procedure complications.
Results:
The implants were successful in all cases, with complete resolution of TR, absence of paravalvular leaks, and adequate transprosthetic gradient. Right ventricular function was preserved, and all patients were discharged 3 to 4 days after the procedure with no significant complications.
Conclusion:
Transcatheter tricuspid valve replacement using the LuX-Valve Plus was shown to be a viable and safe alternative for patients with severe TR and high surgical risk. The use of 3D TEE was essential to the planning and execution of the implant. Further studies are needed to assess durability and long-term clinical outcomes.
976